 The overall three-dimensional shape of a protein molecule is the tertiary structure. The anti-parallel ß-sheet is more stable due to the more well-aligned hydrogen bonds. The two strands can be either parallel or anti-parallel depending on whether the strand directions (N-terminus to C-terminus) are the same or opposite. The carbonyl oxygens in one strand bonds with the amino hydrogens of the adjacent strand. The sheet conformation consists of pairs of strands lying side-by-side. The hydrogen bonding in a ß-sheet is between strands (inter-strand) rather than within strands (intra-strand). 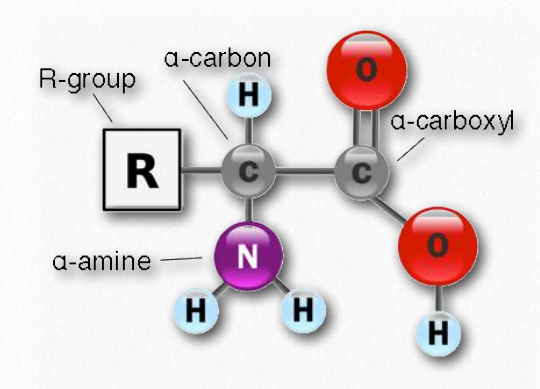
The side-chain substituents of the amino acids fit in beside the N-H groups. The hydrogen bonds make this structure especially stable. Hydrogen bonds form between the oxygen of each C=O bond in the strand and the hydrogen of each N-H group four amino acids below it in the helix. 
The side-chain substituents of the amino acid groups in an α-helix extend to the outside. The α-helix is a right-handed coiled strand. The two main types of secondary structure are the α-helix and the ß-sheet. Stretches or strands of proteins or peptides have distinct, characteristic local structural conformations, or secondary structure, dependent on hydrogen bonding. While the amino acid sequence makes up the primary structure of the protein, the chemical/biological properties of the protein are very much dependent on the three-dimensional or tertiary structure. Often, post-translational modifications, such as glycosylation or phosphorylation, occur which are necessary for the biological function of the protein. Proteins are synthesized by a series of steps called transcription (the use of a DNA strand to make a complimentary messenger RNA strand – mRNA) and translation (the mRNA sequence is used as a template to guide the synthesis of the chain of amino acids which make up the protein). The amino acid sequence of a protein is encoded in DNA. The remaining 10 are called essential amino acids and must be obtained in the diet. Although 20 amino acids are required for synthesis of various proteins found in humans, we can synthesize only ten. These abbreviations are commonly used to simplify the written sequence of a peptide or protein.ĭepending on the side-chain substituent, an amino acid can be classified as being acidic, basic or neutral. Each amino acid has both a one-letter and three-letter abbreviation. The structures of the 20 amino acids commonly found in proteins are shown in Figure 1. These side chains confer different chemical, physical, and structural properties to the final peptide or protein. The amino acids differ in structure by the substituent on their side chains. The terms, amino-terminus and N-terminus, describe the end of the sequence with a free α-amino group. The end of the peptide or protein sequence with a free carboxyl group is called the carboxy-terminus or C-terminus. A protein can be made up of one or more polypeptide molecules. Sequences with fewer than 50 amino acids are generally referred to as peptides, while the terms, protein and polypeptide, are used for longer sequences. This difunctionality allows the individual amino acids to join in long chains by forming peptide bonds : amide bonds between the -NH 2 of one amino acid and the -COOH of another. Amino acids, as their name indicates, contain both a basic amino group and an acidic carboxyl group. :max_bytes(150000):strip_icc()/amino_acid-1b0e0369462e47c6aa53a45d404715ae.jpg)
There are 20 different standard L-α-amino acids used by cells for protein construction. Proteins are macromolecules and have four different levels of structure – primary, secondary, tertiary and quaternary. The term, structure, when used in relation to proteins, takes on a much more complex meaning than it does for small molecules. It will also cover briefly how protein structure can be affected during formulation and some of the analytical methods which can be used both to determine the structure and analyze the stability of the protein. This technical brief aims to give the reader a quick overview of protein structure. Formulation of a protein drug product can be quite a challenge, and without a good understanding of the nature of protein structure and the conformational characteristics of the specific protein being formulated, the results can be ruinous. Increasingly, drug developers are looking to large molecules, particularly proteins, as a therapeutic option. Implantable Drug Delivery Systems and Combination Products Dosage Forms.Solubility & Bioavailability Enhancement. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
